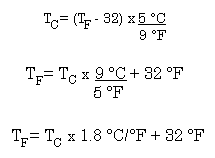
Temperature
Making a relative scale is simple. Create a device which reacts to changes in temperature. Pick one temperature and assign it a number. Next, pick a second temperature and assign it a number. Divide the space between the numbers into regularly sized increments. Now you have a temperature scale.
Celsius is a relative scale. The temperature at which water freezes is defined as 0 °C. The temperature at which water boils is defined as 100 °C. Every temperature is expressed relative to this scale.
Ether boils at 34.6 °C. This value only has meaning because it is a comparison to the temperature at which water freezes and boils.
Fahrenheit is also a relative scale. Science legend has is that 0 °F was the coldest temperature observed and 100 °F was the hottest temperature observed, weather wise, in Dr. Fahrenheit's town.
Kelvin is NOT a relative scale. The Kelvin scale is ABSOLUTE. While studying the relationships between the temperature, the pressure, and the volume of a gas Charles, Boyle and others noted that -273.15 °C must be the lowest temperature attainable. -273.15 °C, or 0 K, is Absolute Zero.
So, the size of the degree is still arbitrary-it is the same size as a degree C. However, 0 K is the lowest possible temperature; it was not arbitrarily assigned.
Temperature Conversions
One conversion is easy. Celsius to Kelvin.
So, just add 273.15 to a temperature given in °C to find the temperature in K.
20 °C = 273.15 K
Or to convert from Kelvin to Celsius subtract 273.15 from a temperature given in K to find the temperature in °C.
196 K = -77 °C
Fahrenheit to Celsius is more complicated. The size of the degree is different, and the zero point is defined differently. You can use a formula:
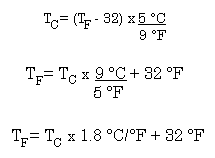
Or you could analyze the temperature scales.
On the celsius scale there are (100 - 0) = 100 degrees between freezing and boiling.On the Fahrenheit scale there are (212 - 32) = 180 degrees between freezing and boiling.
So,
But temperatures cannot be interconverted simply by using this conversion factor. The scales start at different numbers. As an example lets convert the freezing point for benzene from 5.0 °C to °F.5 °C is 5 celcius degrees above the freezing point of water.
5 °C units x 1.8 °F/1 °C = 9 °F units Using the conversion tells us that on the Fahrenheit scale the freezing point of benzene is 9 °F above the freezing point of water; since water freezes at 32 °F, the freezing point of benzene is 41 °F.
During the great oil crisis of the seventies we were all told that we should be cold all winter long; that is, we should keep our thermostats set to 68 °F. What is the temperature in °C?
68 °F is (68 - 32) = 36 °F units above the freezing point of water.36 °F units x 1 °C/1.8 °F = 20 °C units above the freezing point of water. So, 68 °F is 20 °C.
Convert using boiling point instead of freezing point